When it comes to AES methods, the plasma spectroscopies are mainstay of modern day chemistry. 
Inductively coupled plasma atomic emission spectroscopy To detect low concentrations, where only few photons are emitted, photon multiplier can be used. Lastly the detector detects the photons of wavelengths limited by the monochromator that hit its surface. This is done to determine the intensity of different wave lengths throughout the spectrum. 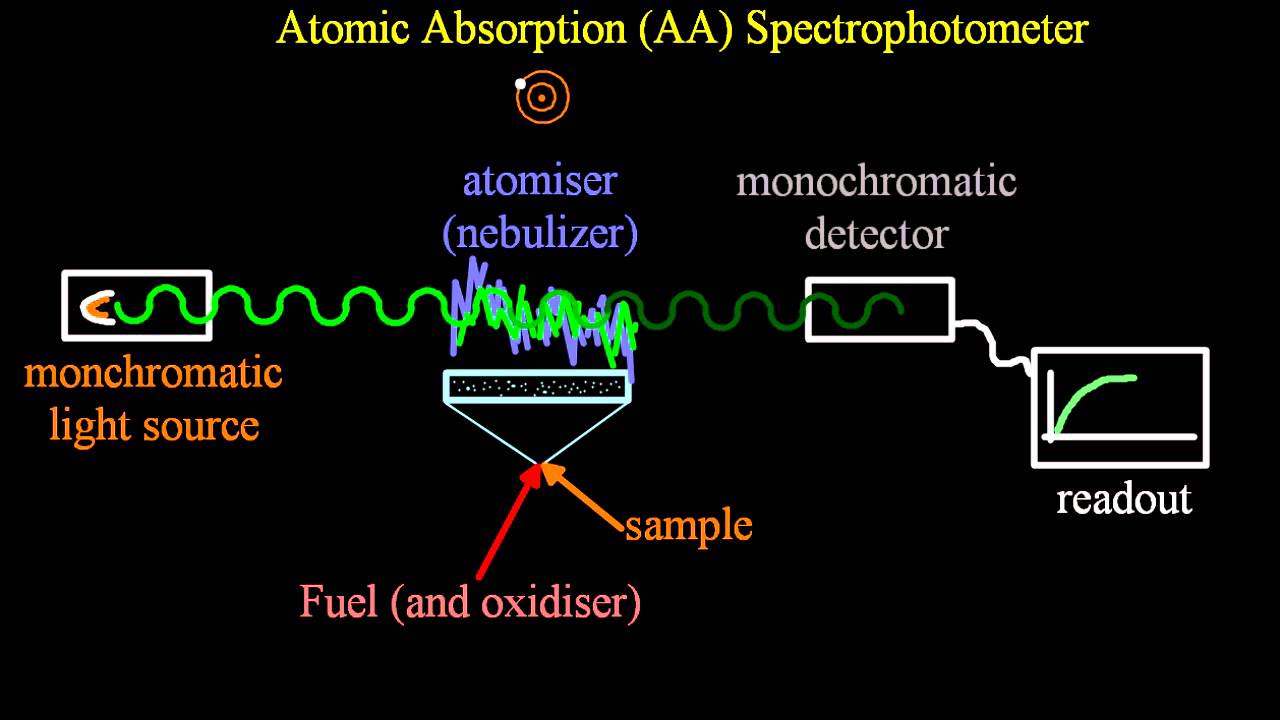
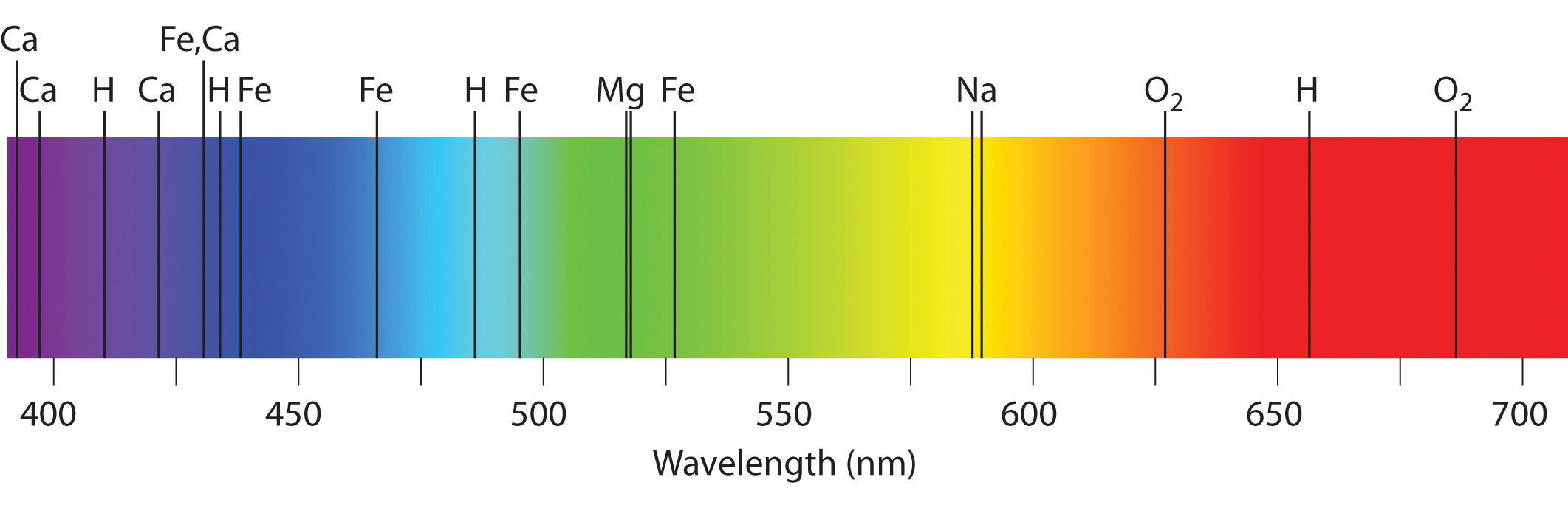
The purpose of monochromator is to limit the radiation emitted from the sample to detector in small wavelength bands. As previously already mentioned plasma is used to excite the atoms of the sample to higher energy level. The AES instrument system typically consists of flame or plasma, monochromator (or polychromator) and detector. Where, ΔE is energy level difference, E 1 and E 2 are the energy levels, h is Planck constant, c is the speed of light and γ is the wavelength. The dependence of energy level difference and wavelength is presented in equation (1). Excitation and decay of an electron, and emission of photon, depicted in Bohr atomic model. Therefore, the element can be detected from the wavelength of the radiation and the concentration of said element can be determined from the intensity of emitted radiation. Since the energy is quantified by the higher and lower energy levels which are specific to each element, the wavelength of the emitted photon is also quantified. This phenomenon is demonstrated in Figure 1. The energy is released as a quantum of electromagnetic radiation, better known as a photon. When decaying, the electron returns from the higher energy level to a lower one and energy is released. The excited state is, however, less stable than the ground state of an atom, this results in decay of the excited state. This energy then excites the electrons in the molecule to a higher energy level. In atomic emission spectroscopy, energy is applied to a molecule in form of light or heat. There are three common methods that follow this phenomenon: flame emission spectroscopy, inductively coupled plasma atomic emission spectroscopy and arc atomic emission spectroscopy. The element can be recognized by the wavelength of the emitted light. Atomic Emission Spectroscopy (AES) is a chemical analysis method in which the quantity of an element is determined by measuring the intensity of light emitted from a flame, plasma, arc or spark.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
